MALARIA ERADICATION
THROUGH VACCINATION
Sanaria's vaccines are intended to be used to prevent malaria in individuals and, in combination with other malaria control measures, to halt transmission of and eliminate malaria from communities.
Sanaria’s PfSPZ Vaccine Provides High-Level Defense Against Malaria In U.S. Navy and Army Clinical Investigation
Sanaria Inc. continues to achieve success in its coordinated efforts to develop a safe, effective and affordable vaccine that delivers mass malaria protection in humans and promotes elimination of the disease worldwide. Research published today in JCI Insight by a team of clinical investigators at the Naval Medical Research Center and Walter Reed Army Institute of Research reports that Sanaria® PfSPZ Vaccine protected against Plasmodium falciparum malaria when clinical trial participants were exposed to two strains of parasites.
Sanaria® PfSPZ Vaccine Against Malaria Receives FDA Fast Track Designation
Sanaria Inc. today announced it has received U.S. Food and Drug Administration (FDA) Fast Track designation for its preventative vaccine for malaria, Sanaria® PfSPZ Vaccine. Sanaria believes its PfSPZ Vaccine is the only malaria vaccine to have ever received this distinction.
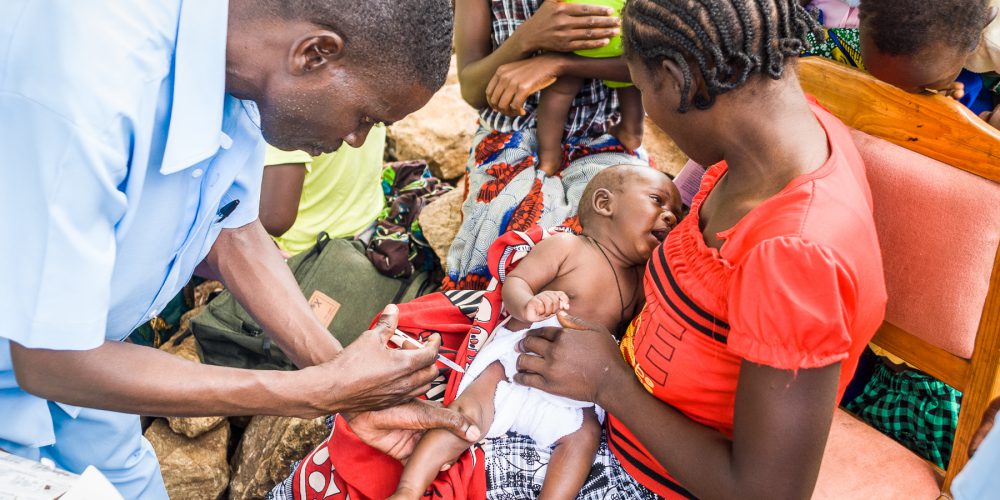
African Trials Moving Ahead with Great Success
Our vaccine is currently being tested for safety, tolerability and efficacy in Mali (West Africa) and Tanzania (East Africa).
Sanaria’s Dr. Stephen L. Hoffman won the Daily Record’s Innovator of the Year Award
Check out more from the Innovator of the Year awards.
In memoriam: Donald ‘DA’ Henderson
Sanaria joins the international public health community in remembering Donald 'DA' Henderson after his death on August 19, 2016. Sanaria was honored to receive a visit from DA in June 2011, where after describing his relentless dedication to the eradication of Smallpox, he offered insights, advice and encouragement to Sanaria at what was a critical time in our PfSPZ Vaccine clinical development program.
Weill Cornell Alumni Association Honors Dr. Stephen Hoffman
This year, Dr. Stephen Hoffman, M.D. ’75 was awarded the Weill Cornell Medicine Alumni Association Award of Distinction for his significant accomplishments as a physician, scientist and entrepreneur. Watch:
An Urgent Need for Malaria Vaccines
Sanaria has an innovative approach to malaria vaccines using Plasmodium falciparum (Pf) sporozoites (SPZ) as the platform technology for immunizing people against malaria infection. The proven effective results of this approach are documented in Sanaria’s publications.
Global Collaboration: The I-PfSPZ Consortium
Semi-annually, Sanaria organizes the international PfSPZ Consortium (i-PfSPZ-C) meeting for our partners, collaborators and funders where we analyze, present and discuss our findings prior to publication. The i-PfSPZ-C allows our collaborators and partners to share their work, modify research and clinical plans based on the consortium efforts and map out future funding needs.
Subscribe
Our Malaria Vaccine Pipeline
Innovative routes to success. A major impact in global health.