MALARIA ERADICATION
THROUGH VACCINATION
Sanaria's vaccines are intended to be used to prevent malaria in individuals and, in combination with other malaria control measures, to halt transmission of and eliminate malaria from communities.
Sanaria Announces the Start of Clinical Trials of its PfSPZ Vaccines in Mali and Indonesia
Sanaria Inc. announces that two new Phase 2 trials of its pioneering malaria vaccines have started. The first is in 6- to 10-year-old children living in Bancoumana, Mali, a malarious region of West Africa. The second is in Indonesian soldiers based in Sumatra, Indonesia. The soldiers will be deploying for six to nine months this coming August to an intensely malarious district in eastern Indonesia.
Plasmodium falciparum 7G8 challenge provides conservative prediction of efficacy of PfNF54-based PfSPZ Vaccine in Africa
Controlled human malaria infection (CHMI) with a Brazilian parasite highly divergent from vaccine and field strains provided stringent estimates of vaccine efficacy in Mali, Africa, and could replace field testing, streamlining vaccine development.
CEO of Sanaria, Dr. Stephen L. Hoffman, to present at the World Vaccine Congress on April 20th, 2022
Dr. Hoffman will be presenting at the World Vaccine Congress on April 20th, 2022.
Virtual Visit from the Logan School
Sanaria virtually hosts group of Colorado students to educate them on vaccines and malaria.
PfSPZ Malaria Vaccine Development Progress by Sanaria in Collaboration with LMIV and MRTC, Highlighted as One of 8 Top NIH Research Projects for 2021
The U.S. National Institutes of Health (NIH) just announced eight top research highlights/projects of 2021 and Sanaria’s PfSPZ-CVac malaria vaccine development efforts in collaboration with the NIH, National Institute of Allergy and Infectious Diseases (NIAID) Laboratory of Malaria, Immunology and Vaccinology (LMIV), and the Malaria Research Training Center (MRTC) in Bamako, Mali is one of the eight. Only two were from NIAID. The other was on COVID-19. This is incredible independent recognition of Sanaria’s progress and the promise of Sanaria’s vaccine program. Learn More
Major Breakthroughs Presented at the International PfSPZ Consortium Meeting
For the second year running, the International PfSPZ Consortium (i-PfSPZ-C) meeting was held as a virtual event on the 9th and 10th December 2021. There were 237 attendees from 70 institutions based in 26 different countries spanning the globe, with colleagues in Australia and Indonesia staying up into the early hours and those on the west coast of the USA rising early to make their contributions to a packed and exciting agenda.
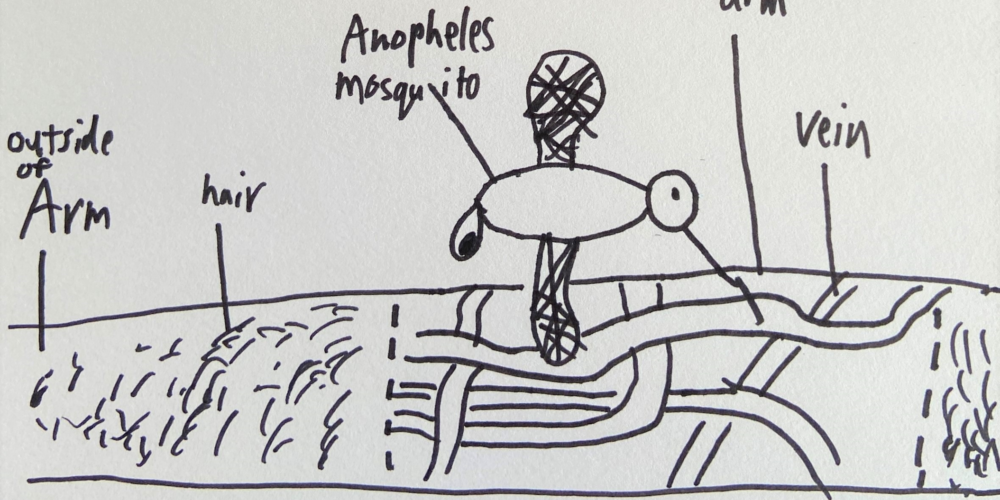
An Urgent Need for Malaria Vaccines
Sanaria has an innovative approach to malaria vaccines using Plasmodium falciparum (Pf) sporozoites (SPZ) as the platform technology for immunizing people against malaria infection. The proven effective results of this approach are documented in Sanaria’s publications.
Global Collaboration: The I-PfSPZ Consortium
Semi-annually, Sanaria organizes the international PfSPZ Consortium (i-PfSPZ-C) meeting for our partners, collaborators and funders where we analyze, present and discuss our findings prior to publication. The i-PfSPZ-C allows our collaborators and partners to share their work, modify research and clinical plans based on the consortium efforts and map out future funding needs.
Subscribe
Our Malaria Vaccine Pipeline
Innovative routes to success. A major impact in global health.